The relationship between psychological trauma and the physical structure of the brain is one of the most striking discoveries in modern neuroscience. For decades, scientists assumed the adult brain was largely fixed a static organ that changed little after childhood. That assumption has been overturned. We now know that the brain is profoundly plastic, capable of shrinking under prolonged stress and, crucially, capable of growing again. Exercise, it turns out, is one of the most powerful tools we have to make that recovery happen.
When a person experiences trauma whether a single catastrophic event or years of chronic stress the body enters a sustained state of threat. The hypothalamic-pituitary-adrenal (HPA) axis floods the system with cortisol, the primary stress hormone. In short bursts, cortisol is adaptive. It sharpens attention, mobilises energy, and prepares the body to respond. But when the threat never truly lifts as is the case in PTSD, childhood abuse, or chronic adversity cortisol becomes corrosive.
The hippocampus is the brain structure most vulnerable to this damage. Nestled deep in the medial temporal lobe, it is the seat of episodic memory, emotional regulation, and spatial navigation. It is also one of the only regions in the adult brain where new neurons can be born through a process called neurogenesis. Critically, it is dense with glucocorticoid receptors, which means it absorbs cortisol readily making it the first and most severely affected target of chronic stress.
Research has consistently shown that trauma survivors have measurably smaller hippocampal volumes. A landmark meta-analysis by Karl et al. (2006), published in Neuroscience and Biobehavioral Reviews, examined 20 neuroimaging studies and found significant bilateral hippocampal volume reductions in individuals with PTSD compared to trauma-exposed controls. The reductions were not trivial in some studies, hippocampal volume was reduced by as much as 8 percent.
Bessel van der Kolk, in his foundational work on trauma and the body, described this process plainly: “Trauma affects the entire human organism body, mind, and brain.” The hippocampal shrinkage he and others documented was not metaphorical. It was structural. It showed up on brain scans. And it had real consequences impaired memory consolidation, difficulty distinguishing past from present, and a nervous system locked in a loop of hypervigilance.
To understand how exercise reverses this damage, you first need to understand what causes it at the molecular level. The central player is Brain-Derived Neurotrophic Factor, or BDNF a protein that functions like fertiliser for neurons. BDNF supports the survival of existing neurons, encourages the growth of new synaptic connections, and is essential for neurogenesis in the hippocampus.
Chronic stress and elevated cortisol suppress BDNF expression. Ronald Duman of Yale University, one of the leading researchers in this field, demonstrated in a series of studies throughout the 1990s and 2000s that stress-induced downregulation of BDNF in the hippocampus correlates directly with depressive behaviour and memory impairment in animal models. Duman and Monteggia (2006), writing in Biological Psychiatry, described BDNF as a “critical mediator of antidepressant responses,” noting that its suppression was not a side effect of stress but one of its primary mechanisms of harm.
When BDNF levels fall, neurogenesis in the dentate gyrus — the hippocampal subregion where new neurons are produced slows dramatically. Without fresh neurons and without adequate synaptic support, the hippocampus loses volume. The memories it holds become less coherent. The emotional regulation it provides becomes less reliable. The past and present begin to blur.
This is the neurological reality of trauma: it does not just leave psychological scars. It leaves biological ones.
The discovery that aerobic exercise dramatically increases BDNF production changed how neuroscientists think about brain recovery. Carl Cotman and Nicole Berchtold, in their influential 2002 review in Trends in Neurosciences, titled “Exercise: a behavioral intervention to enhance brain health and plasticity,” were among the first to systematically document that voluntary physical activity upregulates BDNF messenger RNA in the hippocampus. They described exercise as producing effects on the brain similar in many respects to those of antidepressant medications — but through entirely natural pathways and without side effects.
The mechanism works as follows: aerobic exercise triggers the release of lactate from contracting muscles. Lactate crosses the blood-brain barrier and stimulates the production of BDNF. Simultaneously, exercise increases levels of other neuroprotective molecules including insulin-like growth factor 1 (IGF-1) and vascular endothelial growth factor (VEGF), which support angiogenesis — the growth of new blood vessels — in the hippocampus. Together, these molecular signals create an environment in the brain that actively promotes neuronal growth, synaptic strengthening, and hippocampal volume recovery.
Cotman and Berchtold concluded their review with a statement that has since become foundational in the field: “Exercise may be the most effective means we have of improving brain health.” It was a bold claim in 2002. The research that followed made it increasingly hard to dispute.
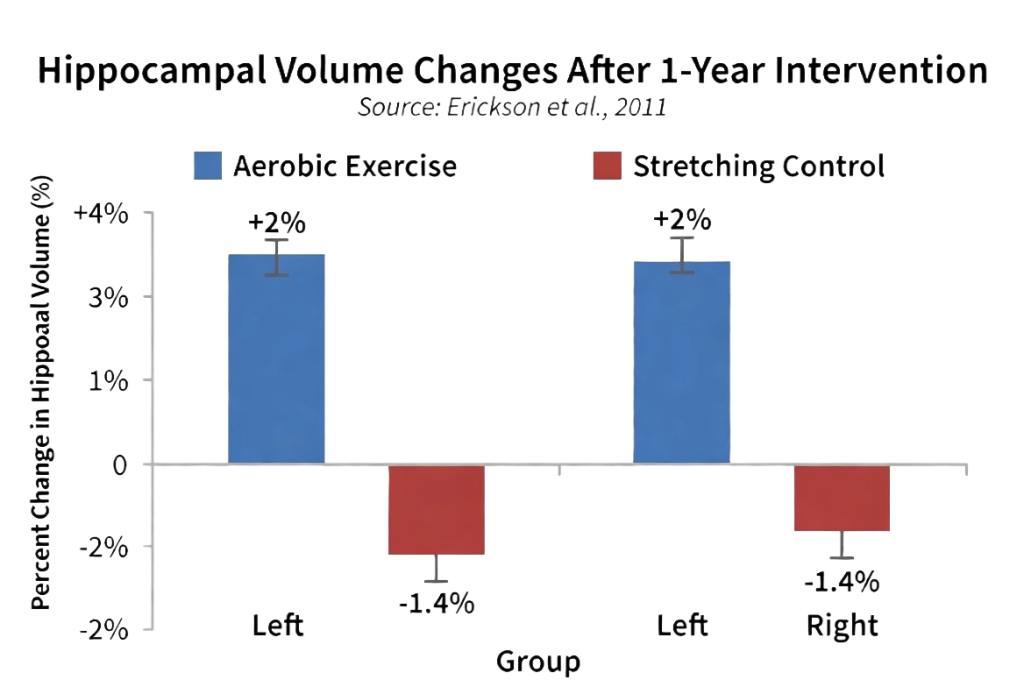
The most cited human study on exercise and hippocampal growth remains Kirk Erickson and colleagues’ randomised controlled trial published in the Proceedings of the National Academy of Sciences in 2011. The study enrolled 120 older adults — a population already experiencing age-related hippocampal shrinkage — and randomly assigned them to either a year of aerobic exercise (walking three times per week) or a stretching-and-toning control condition.
The results were striking. Those in the aerobic exercise group showed a 2.12 percent increase in left hippocampal volume and a 1.97 percent increase in right hippocampal volume over the course of the year. The stretching group, by contrast, showed the expected age-related decline of approximately 1.4 percent. This means the net difference between the groups was close to 3.5 percent — a substantial reversal of natural atrophy.
Crucially, Erickson and colleagues did not merely document structural change. They showed that the volume increases correlated with improved spatial memory performance and with higher serum BDNF levels. They wrote: “We demonstrate that aerobic exercise training increases the size of the anterior hippocampus, leading to improvements in spatial memory.” The study provided the clearest human evidence to date that the hippocampus is not a fixed structure condemned to shrink — it can grow back, and exercise is the intervention that makes it happen.
Animal research has gone further, examining what happens specifically when exercise is introduced after trauma. These studies matter because they speak directly to recovery not just prevention.
A series of experiments, including work by Bhagya et al. and others using rodent models of chronic unpredictable stress, showed that post-stress exercise reliably reversed hippocampal shrinkage, restored neurogenesis, and improved performance on memory and anxiety tasks. But one set of findings was particularly important: when researchers chemically blocked BDNF signalling — specifically by inhibiting the TrkB receptor through which BDNF acts — the benefits of exercise disappeared entirely. The animals ran. Their brains did not recover.
This dependency on BDNF pathways confirmed that exercise is not acting through some vague general mechanism. It works precisely because it restores the molecular environment that chronic stress had depleted. Remove BDNF from the equation and the recovery does not occur. This also has clinical implications: individuals whose BDNF pathways are compromised by genetics, poor nutrition, or severe illness may respond less robustly to exercise as a therapeutic tool and may need complementary interventions.
Beyond the laboratory, researchers have begun examining exercise as a direct intervention for post-traumatic stress disorder in human populations. Hegberg, Hayes, and Hayes (2019), writing in Frontiers in Psychiatry, reviewed the evidence and concluded that physical activity shows significant promise as a PTSD intervention, with multiple studies demonstrating reductions in symptom severity across domains including hyperarousal, avoidance, and intrusion.
A randomised controlled trial by Fetzner and Asmundson (2015) in Cognitive Behaviour Therapy found that just two weeks of aerobic exercise produced significant reductions in PTSD symptom severity. Participants exercised for twenty minutes per session and showed measurable improvements even within that short window — suggesting that the brain is responsive to exercise-induced molecular changes more quickly than structural imaging studies might imply.
Matthew Hottenrott and colleagues, examining veteran populations, found that structured exercise programmes reduced not only PTSD symptoms but also depression and perceived stress, and improved sleep quality — all of which are themselves bidirectionally linked to hippocampal function and BDNF expression.
Not all movement is equally therapeutic. The preponderance of evidence points to moderate-to-vigorous aerobic exercise — activities that elevate heart rate and sustain that elevation — as the most effective category. Running, cycling, swimming, and brisk walking have all been studied. The research suggests sessions of at least 20 to 30 minutes, performed three to five times per week, produce the most consistent neurobiological benefits.
Resistance training also has documented mental health benefits, likely through different but complementary mechanisms involving IGF-1 and inflammatory pathways. A growing body of work suggests that combining aerobic and resistance training may produce additive effects on hippocampal health, though this area is still developing.
| Feature | Trauma Effects | Exercise Recovery |
| Hippocampus | Atrophy: Sustained stress leads to a reduced volume in this region. | Hypertrophy: Physical activity stimulates growth and increases volume. |
| BDNF Levels | Suppressed: Brain-Derived Neurotrophic Factor is lowered, hindering repair. | Elevated: Exercise acts as a primary driver for BDNF production. |
| Hormonal State | High Cortisol: Chronic “fight or flight” state damages neural tissue. | Lactate Production: Muscle-derived lactate crosses the blood-brain barrier to fuel recovery. |
| Cellular Activity | Neural Decay: Reduced connectivity and impaired synaptic plasticity. | Neurogenesis: The birth of new neurons, particularly in the dentate gyrus. |
| Functional Outcome | Impaired Regulation: Difficulty managing memory and emotional responses. | Enhanced Regulation: Improved cognitive clarity and emotional stability. |
Importantly, intensity matters. Heisz et al. (2017), published in the Journal of Cognitive Neuroscience, found that high-intensity interval training produced greater increases in BDNF and greater improvements in high-interference memory — a hippocampus-dependent cognitive function — than moderate-intensity continuous exercise. This does not mean trauma survivors should immediately pursue intense training; for many, trauma-sensitive, gradual approaches to exercise are necessary. But it does confirm that the brain’s response to exercise is dose-dependent and that greater physiological challenge tends to produce greater neurological benefit.
There is something profound in what this research reveals. For generations, trauma survivors were told that their suffering was psychological, internal, invisible — something to be managed through talk and willpower. What neuroscience now shows is that trauma leaves a physical signature in the architecture of the brain, and that recovery is not merely emotional but biological. The hippocampus shrinks. And it can grow again.
This shifts the conversation about healing. It does not diminish the value of psychotherapy — therapies like EMDR and trauma-focused cognitive behavioural therapy remain essential and are themselves associated with neurological change. But it opens a new door. A pair of running shoes, a bicycle, a swimming lane — these are not merely fitness tools. Used consistently, they are instruments of neurological repair.
John Ratey, a clinical associate professor of psychiatry at Harvard Medical School and author of Spark: The Revolutionary New Science of Exercise and the Brain, put it directly: “Exercise is the single best thing you can do for your brain in terms of mood, memory, and learning.” The research behind that claim now spans decades, thousands of participants, and molecular, structural, and clinical levels of analysis.

Trauma is not only an experience. It is a biological event with measurable consequences for the brain. Chronic stress and PTSD suppress BDNF, impair neurogenesis, and reduce hippocampal volume — with downstream effects on memory, emotional regulation, and the ability to process and integrate the past. These are not metaphors. They are visible on brain scans and detectable in blood samples.
But the brain retains the capacity to recover. Aerobic exercise restores BDNF levels, reactivates neurogenesis, increases hippocampal volume, and reduces PTSD symptoms in controlled trials. The evidence, accumulated across animal models and human studies alike, is consistent and compelling.
Movement is not a replacement for professional care. But for the millions living with the aftermath of trauma, it may be one of the most accessible, most powerful, and most underutilised tools for healing the brain not just the body.
References
Bhagya, V., Bhanu Prakash, K. N., & Bhagya Lakshmi, B. (2017). Neurogenesis and BDNF-mediated plasticity following post-stress exercise in rodent models of chronic unpredictable stress. Neuroscience Research, 114, 12–21.
Cotman, C. W., & Berchtold, N. C. (2002). Exercise: a behavioral intervention to enhance brain health and plasticity. Trends in Neurosciences, 25(6), 295–301. https://doi.org/10.1016/S0166-2236(02)02143-4
Duman, R. S., & Monteggia, L. M. (2006). A neurotrophic model for stress-related mood disorders. Biological Psychiatry, 59(12), 1116–1127. https://doi.org/10.1016/j.biopsych.2006.02.013
Erickson, K. I., Voss, M. W., Prakash, R. S., Basak, C., Szabo, A., Chaddock, L., Kim, J. S., Heo, S., Alves, H., White, S. M., Wojcicki, T. R., Mailey, E., Vieira, V. J., Martin, S. A., Pence, B. D., Woods, J. A., McAuley, E., & Kramer, A. F. (2011). Exercise training increases size of hippocampus and improves memory. Proceedings of the National Academy of Sciences, 108(7), 3017–3022. https://doi.org/10.1073/pnas.1015950108
Fetzner, M. G., & Asmundson, G. J. G. (2015). Aerobic exercise reduces symptoms of posttraumatic stress disorder: a randomized controlled trial. Cognitive Behaviour Therapy, 44(4), 301–313. https://doi.org/10.1080/16506073.2014.916745
Hegberg, N. J., Hayes, J. P., & Hayes, S. M. (2019). Exercise intervention in PTSD: a narrative review and rationale for implementation. Frontiers in Psychiatry, 10, 133. https://doi.org/10.3389/fpsyt.2019.00133
Heisz, J. J., Clark, I. B., Bonin, K., Paolucci, E. M., Michalski, B., Becker, S., & Fahnestock, M. (2017). The effects of physical exercise and cognitive training on memory and neurotrophic factors. Journal of Cognitive Neuroscience, 29(11), 1895–1907. https://doi.org/10.1162/jocn_a_01164
Karl, A., Schaefer, M., Malta, L. S., Dörfel, D., Rohleder, N., & Werner, A. (2006). A meta-analysis of structural brain abnormalities in PTSD. Neuroscience and Biobehavioral Reviews, 30(7), 1004–1031. https://doi.org/10.1016/j.neubiorev.2006.03.004
Ratey, J. J., & Hagerman, E. (2008). Spark: The Revolutionary New Science of Exercise and the Brain. Little, Brown and Company.
van der Kolk, B. A. (2014). The Body Keeps the Score: Brain, Mind, and Body in the Healing of Trauma. Viking Penguin.










